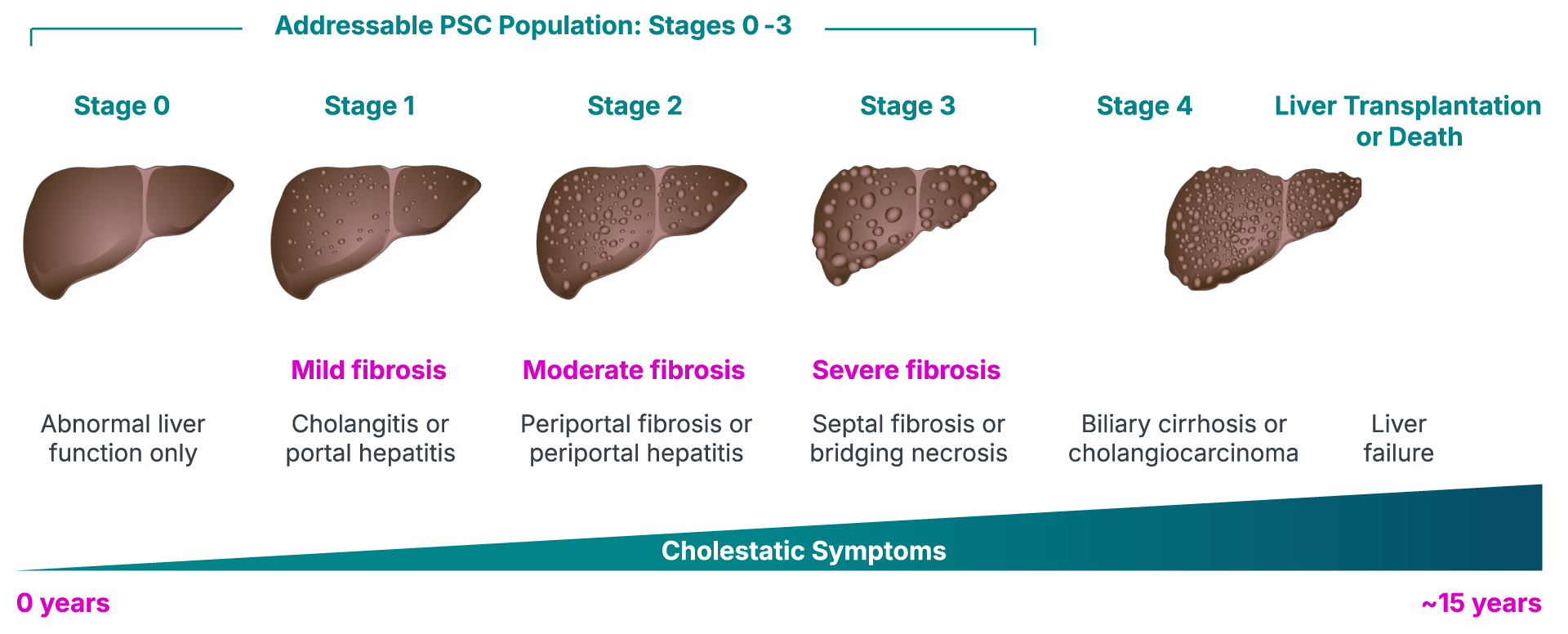
Innovative Portfolio & Pipeline

The Esperion next-generation ACLY inhibitor discovery program is a world-class approach

ACLY Inhibitor Discovery Program Objectives
 Use leading edge approaches and technology to identify/design differentiated new chemical entities
Use leading edge approaches and technology to identify/design differentiated new chemical entities Improve potency and selectivity several orders of magnitude vs. current active site inhibitors
Improve potency and selectivity several orders of magnitude vs. current active site inhibitors Target several cell types important in disease pathogenesis
Target several cell types important in disease pathogenesis Identify potential indications and patient populations using leading edge bioinformatic approaches
Identify potential indications and patient populations using leading edge bioinformatic approaches
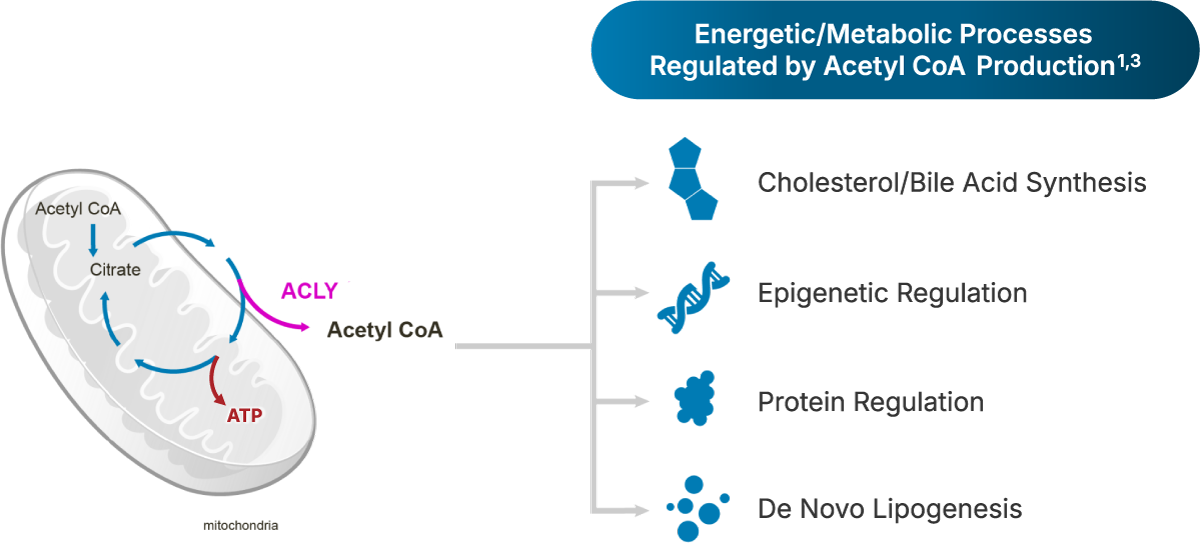
ACLY is the key enzyme in the non-canonical TCA cycle, an energy nexus in all cells1,2
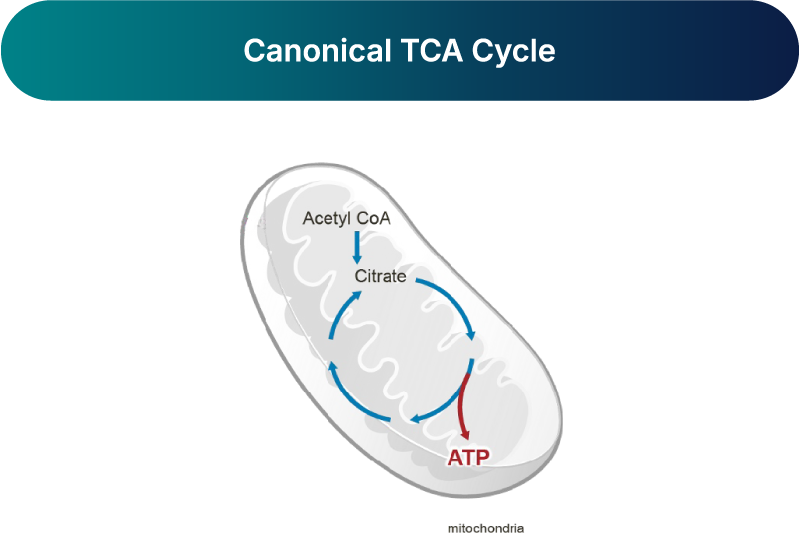
The canonical TCA cycle, also known as the Krebs Cycle, is primarily a catabolic pathway that consumes lipids and carbohydrates to generate ATP for energy production and occurs in mitochondria.1

The non-canonical TCA cycle is primary an anabolic path-way that supports cell states with high demands due to accelerated proliferation and/or effector functions; is utilized by highly proliferative cells, cancer cells, and stem cells; and occurs in mitochondria, cytoplasm, and nucleus.1,2
Allosteric ACLY inhibition is a novel approach to targeting this key enzyme

Active site ACLY inhibitors prevent the substrate (coenzyme A) from binding, thus inhibiting enzyme activity

Allosteric ACLY inhibitors bind allosteric sites distant from the active site which changes the conformation of the active site, thus inhibiting enzyme activity.
Investigating potential advantages of allosteric inhibition